Diatoms, a group of tiny algae, are also known as “living opals” because of the strange, beautiful properties of their silica shells. But what genes are responsible for such mesmerizing exteriors? Setsuko Wakao and Kris Niyogi, biologists at both UC Berkeley and Lawrence Berkeley National Laboratory, aim to find out.
The Joint Genome Institute presents the Genome Insider podcast.
Transcript of the episode
ALISON: Hey! I’m Alison Takemura, and this is Genome Insider, a podcast of the US Department of Energy Joint Genome Institute, or JGI. In soils, waterways, and the ocean, there is an exquisite group of microscopic organisms. They’re called diatoms.
SETSUKO: There are many, many different species. And what’s really amazing about them is that they’re very beautiful. They’re very beautiful in terms of the larger shape of the cell but also when you look at them under the microscope, they have very intricate patterns at the micro- and nanoscale.
ALISON: That’s Setsuko Wakao, a research scientist at UC Berkeley and Lawrence Berkeley National Lab.
And Setsuko is studying diatoms. Diatoms make, or biomineralize, hard, silica shells. And these shells give them some unique shapes — like stars, triangles, and pinwheels, bean pods, orange slices, and saucers. I know. Weird right?
Also, did you know that it was a Victorian hobby to arrange diatoms into pleasing symmetrical patterns? I didn’t!
But even stranger than what people do with them is that the shells of these unusual organisms have nanoscale patterns. And the patterns create colors. Their shells’ nanostructure affects the behavior of photons. You’re actually already probably familiar with this effect. It’s the same thing you see in peacock feathers and butterfly wings. They’re so beautifully vibrant, not because of pigments, but because of physical structures invisible to the naked eye. Same thing with diatoms. Their shells’ funky interactions with light have earned diatoms the nicknames “jewels of the sea” and “living opals.”

Diatoms arranged in a symmetric pattern, like the Victorians used to do back in the day. (W.M. Grant)
But diatoms are more than just their good looks. They’re self-sufficient. They make food from sunlight using photosynthesis. And in the ocean, diatoms have been wildly successful; they make up about half of the ocean’s photosynthesizing microbial community: the phytoplankton. It’s a wonderfully evocative name: “phyto” means plant, and “plankton” means wanderer.
As diatoms wander, they have a huge effect on our planet. They account for 20 percent of the carbon fixed globally every year. They transform carbon dioxide into biomass that they can incorporate into their bodies.
And then when they die, their shells are heavy enough that they can cause them to sink to their watery graves. In death, diatoms take some of the carbon that they’ve fixed with them. And it becomes locked away.
KRIS: And so this is a, an important mechanism for sequestering carbon in the deep ocean over geological time, those algae that grew millions of years ago, can be turned into oil reserves. And so essentially, what we’re doing now by burning those oil reserves is releasing carbon dioxide that was fixed by phytoplankton many millions of years ago, all at once.
ALISON: That’s Kris Niyogi, a plant and microbial biologist, and like Setsuko, also affiliated with UC Berkeley and Lawrence Berkeley National Lab. Setsuko is a member of Kris’ lab.

The light green swirls in the sea are phytoplankton, visible from space. This bloom occurred off the coast of western Iceland. (NASA Goddard Space Flight Center, CC BY 2.0)
Kris and Setsuko are interested in better understanding this process where diatoms sink and sequester carbon. It’s part of a phenomenon called the “biological carbon pump” — think of it as diatom deaths and their heavy silica shells “pumping” or moving carbon from one place to another.
KRIS: I’m really interested in what sort of the, the underlying fundamental mechanisms are. This is something that’s really important for the biological carbon pump, and we need to understand how they do this silica biomineralization to fully understand how that process works.
ALISON: So, diatoms affect the Earth’s carbon cycle. But …
SETSUKO: As important as it is, diatoms themselves and the biosynthesis of these silica cell walls are still not understood completely in terms of what genes are involved.
ALISON: And so, Setsuko and Kris are investigating how diatoms make their shells.
The answers could influence the design and manufacture of new materials, as well as give us insight into a natural process of sequestering carbon.
But first, let’s get to know diatoms a little better.
ALISON: Can you describe what a diatom might look like?
SETSUKO: They’re almost like a mesh of silica patterns.
ALISON: I tried to imagine what that might look like, and thought: maybe chainmail? Eh, no.
SETSUKO: Well, it’s not really like a chain— links of chains. Because it’s more like if you have some kind of contiguous surface, and you started poking holes in a very regular pattern, so [there isn’t this kind of,] it’s still two dimensional. But the pores are formed in very regular ways that makes it look like a mesh.
ALISON: It’s kind of like a tea strainer. Scientists think that diatoms are so abundant, in part, because their silica cell walls are made with the element silicon (Si) instead of carbon.
SETSUKO: So if you’re not using carbon that you fixed, but rather a different element, you’re being a little bit more efficient in terms of your energy, because you can use that carbon that you fixed for some other parts of the cell. The other advantage that is commonly discussed is that by having a silica cell wall, it’s very hard to be digested.

Kris Niyogi, plant and microbial biologist at UC Berkeley and Lawrence Berkeley National Lab. (Peter Barreras)
So when you think about the marine environment, where there are predators that eat up all these phytoplankton, maybe it allowed them to survive better. And there are other points like their optical properties and how that might help capture more light.
ALISON: But that’s still kind of iffy. What’s clear is these shells have a powerful influence on their lives. So, how do they make them? Right now, a little is known. It seems like there’s a protein template that the silica structure gets built upon. But Setsuko and Kris aim to dig deeper — to figure out what genes are involved.
So they’re coming at the problem, cleverly, from the side. They’re analyzing not only diatoms, but a related group of microalgae called synurophytes. Their name means a plant with a tail. They have flagella, which look like little tails.
SETSUKO: So these are algae that grow in freshwater. They also make silica cell walls but in very different forms.
ALISON: Unlike diatoms, with their perforated mesh shells, synurophytes make scales. And they overlap, just like on the body of a fish.
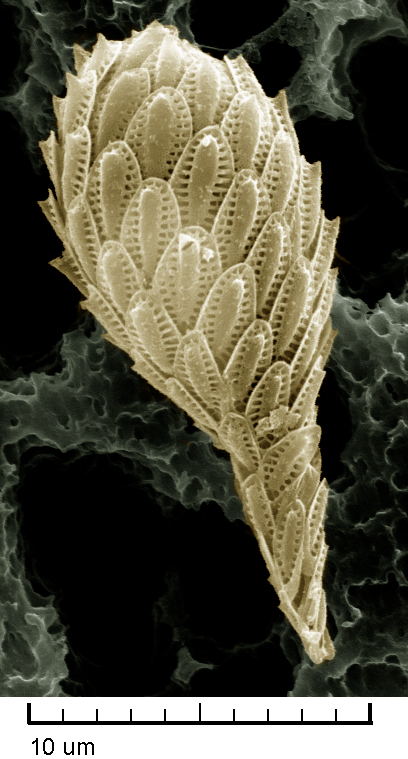
A synurophyte, Synura petersenii, with its silica scales. False-colored. (Drew Lindow, CC BY-SA 3.0)
SETSUKO: They become completely covered in these scales.
ALISON: And these scales are actually quite big relative to the synurophyte’s body. To me they look like a ball wrapped in leaves. Anyway, Setsuko and Kris think synurophytes and their genes could give them clues to how diatoms make their hard shells.
SETSUKO: If we sequence the genomes of these synurophytes, we might find common genes that will tell us about the important processes in silica cell wall biosynthesis by comparing it with diatoms.
ALISON: So, Setsuko and Kris are working on sequencing synurophyte genomes with JGI. And, in the meantime, they’re poring over already sequenced diatom genomes. JGI actually sequenced the very first diatom genome from the species Thalassiosira pseudonana. And since then, JGI has sequenced four more diatoms. And it recently committed to sequence an additional one hundred diatom genomes. Setsuko and Kris will be able to use all of that data.
But comparing just genome sequences is a static enterprise. In other words, it’s limited. Setsuko and Kris also want to investigate genomes dynamically. So, they plan to figure out which genes synurophytes express when they make their silica scales. Because they don’t make them all the time.
SETSUKO: What’s unique about synurophytes, is that they’re not completely dependent on silica as diatoms are.
ALISON: Diatoms can’t grow without silica, because they can’t grow without their cell walls.
SETSUKO: So, diatoms, if you remove silica completely, they can’t grow. Which makes it difficult for us as an experimental system, because we would like to compare when you don’t have silica versus when you have a lot of silica, but you cannot do this with diatoms.
ALISON: So, Setsuko and Kris are looking to the synurophyte cousins for answers. Synurophytes do grow without silica. They’re perfectly OK being unprotected, naked cells. And that flexibility gives scientists an opportunity.
SETSUKO: You can compare the genes that are expressed when it doesn’t make any scales, versus when you dump an amount of silica on it. And now it’s producing this multiple layers of scales.
ALISON: Setsuko and Kris aim to find what genes get switched on when synurophytes go into these scale-making mode. Those genes are likely to be important for building the silica (SiO2) cell wall. So, the team will then look for those genes in diatoms.
SETSUKO: And the last part is to test whether disrupting those genes actually lead to the loss or alteration of silica cell walls.
ALISON: That’s how the team will make sure they’ve found the right genes. They’ll use a technique that’s growing in popularity: CRISPR interference, or CRISPRi. It blocks the expression of genes using small CRISPR guide RNA sequences that target the genes of interest. And the JGI is making the DNA templates for those guide RNA sequences.
SETSUKO: So, we’re taking advantage of the DNA synthesis capacity of JGI to make CRISPRi guide RNAs to test all these candidate genes.
ALISON: In other words, Setsuko and Kris will see whether blocking the expression of these genes in a model diatom actually changes its cell wall. The approach is like having workers at a construction site. Let’s say, some of them are building a brick wall. But you don’t know who. You do have a list of people, though. So you do an experiment. You prevent one worker at a time from going to the job site. And then you see whose absences affect the wall the most. Is it not being built well or even at all anymore? That’s basically what Kris and Setsuko are going to try to do with the diatom genes.
And they hope that all of these efforts — analyzing synurophyte genomes, depriving them of silica, and blocking the expression of interesting genes — will help illuminate how diatoms build their silica shells.

Zooming in on a marine diatom, Detonula pumila, you can see the mesh-like nanoscale patterning on its shell surface. (Health Sciences and Nutrition, CSIRO)
One potential result of this knowledge? It could help researchers develop new biomaterials. These biomaterials could be based on a diatom shell’s nanoscale patterns.
SETSUKO: So, all these very regular and small patterns lead to certain optical properties.
ALISON: And those properties could be useful for biomedical or electronic applications, for example. The advantages of a biomaterial?
SETSUKO: A biomaterial is very reliable. I mean, biology can do things easily, as compared to chemical processes.
ALISON: Those processes require hard work in the lab. Diatoms, by contrast, just need sunlight, water, and a few trace nutrients to build a nanomaterial.
But that’s not all that could come from better understanding how diatoms biomineralize their cell walls. Even though it’s a tiny, molecular process, it could also help us fight climate change.
KRIS: I think it’s far in the future. But understanding how biomineralization works could lead to new ideas for, for carbon sequestration.
ALISON: Setsuko says that one example of how not to do it is to add silica into the ocean, in the hope of making more diatoms grow and sink. That could have unanticipated, and undesirable, side effects. Ocean ecology is complex. And adding silica might change the rate of diatom sinking. We could end up sequestering less carbon, which is the opposite of what we want. So, Setsuko and Kris hope that better understanding diatoms will give humans better ideas in the future.
Together, diatoms and their evolutionary cousins, the synurophytes, might show us the way. If some come out of their shells.
ALISON: This episode was directed and produced by me, Alison Takemura, with editorial and technical assistance from Massie Ballon and David Gilbert.
Genome Insider is a production of the Joint Genome Institute, a user facility of the US Department of Energy Office of Science. JGI is located at Lawrence Berkeley National Lab in beautiful Berkeley, California.
A huge thanks to Kris Niyogi and Setsuko Wakao from UC Berkeley and Lawrence Berkeley National Lab’s Biosciences Area’s Molecular Biophysics and Integrated Bioimaging Division for sharing their research and the startling beauty of diatoms. Setsuko is the principal investigator on this project, as part of JGI’s New Investigator program.
If you enjoyed Genome Insider and want to help others find us, leave us a review on Apple Podcasts, Google Podcasts, or wherever you get your podcasts. If you have a question or want to give us feedback, Tweet us @JGI, or record a voice memo and email us at JGI dash comms at L-B-L.gov. That’s jgi dash c-o-m-m-s at l-b-l dot g-o-v.
And because we’re a user facility, if you’re interested in partnering with us, we want to hear from you! We have projects in genome sequencing, synthesis, transcriptomics, metabolomics, and natural products in plants, fungi, algae, and microorganisms. If you want to collaborate, let us know!
Find out more at jgi.doe.gov forward slash user dash programs.
And if you’re interested in hearing about cutting-edge research in secondary metabolites, also known as natural products, then check out JGI’s other podcast, Natural Prodcast. It’s hosted by Dan Udwary and me. That’s it for now. See ya next time!
Additional information related to the episode
- Community Science Program proposal: Sequencing 100 Diatom genomes
- JGI News Release: Tracking Antartic Adaptations in Diatoms
- JGI News Release: First diatom genome sequenced
- Algal genomics resource, Phycocosm
- JGI’s Fungal & Algal Program
- The JGI Strategic Plan, noting an algal focus, is available here
- Our contact info:
- Twitter: @JGI
- Email: jgi-comms at lbl dot gov

